Volatile higher silanes are low temperature, high deposition rate precursors for:
- amorphous hydrogenated silicon (a-Si:H) films
- germanium-doped (compression-strained) silicon
- carbon-doped (tensile-strained) silicon
- silicon CVD, ALD, ALE
- silicon and silicon-binary quantum-dots by photolytic or laser induction
- silicon-based photovoltaics
By appropriate selection of the higher silane precursor and deposition conditions, silicon deposition can be shifted from amorphous hydrogenated silicon toward microcrystalline silicon structures.
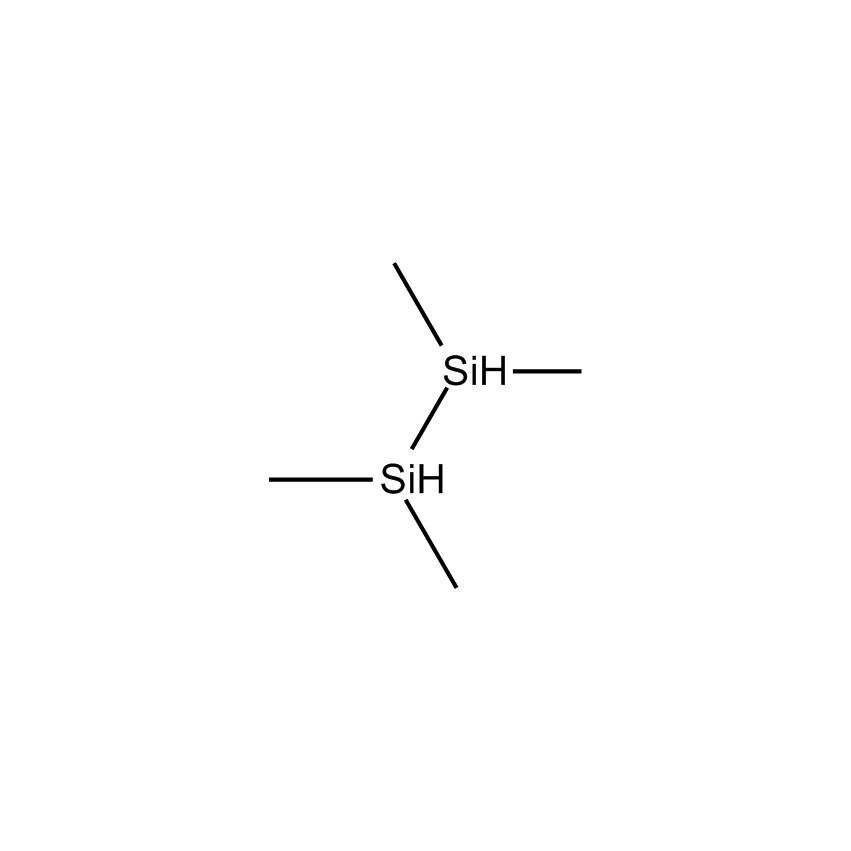
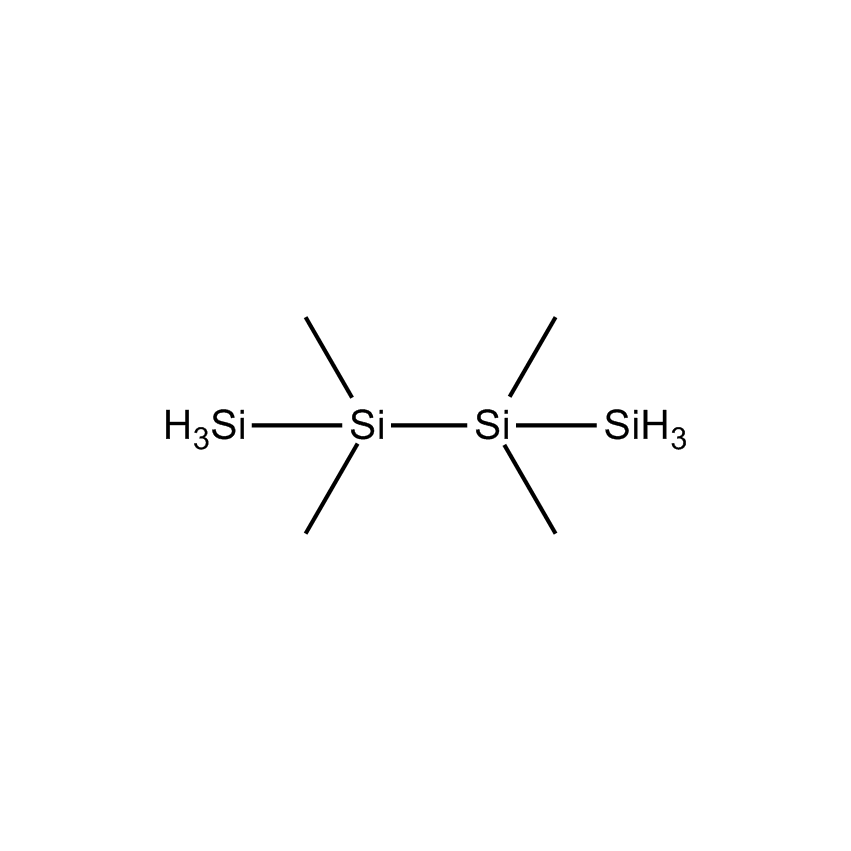
Volatile higher silanes containing three or more silicon atoms and hydrogen are more accurately termed perhydridooligosilanes. They are volatile pyrophoric liquids with the general formula SinH2n+2, which may be depicted as seen below.
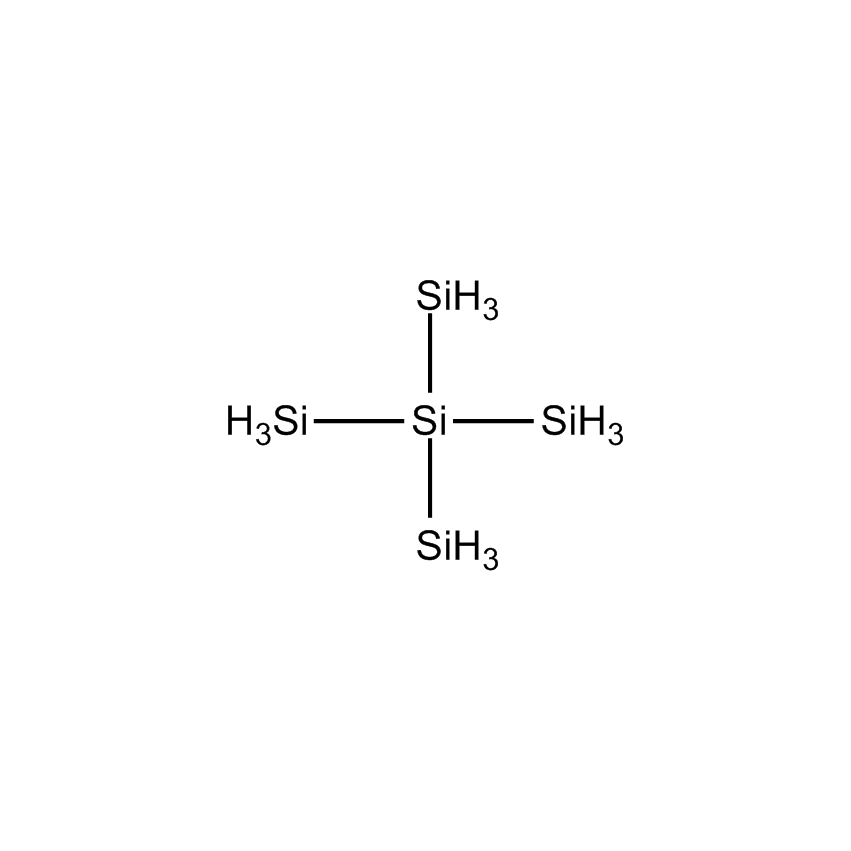
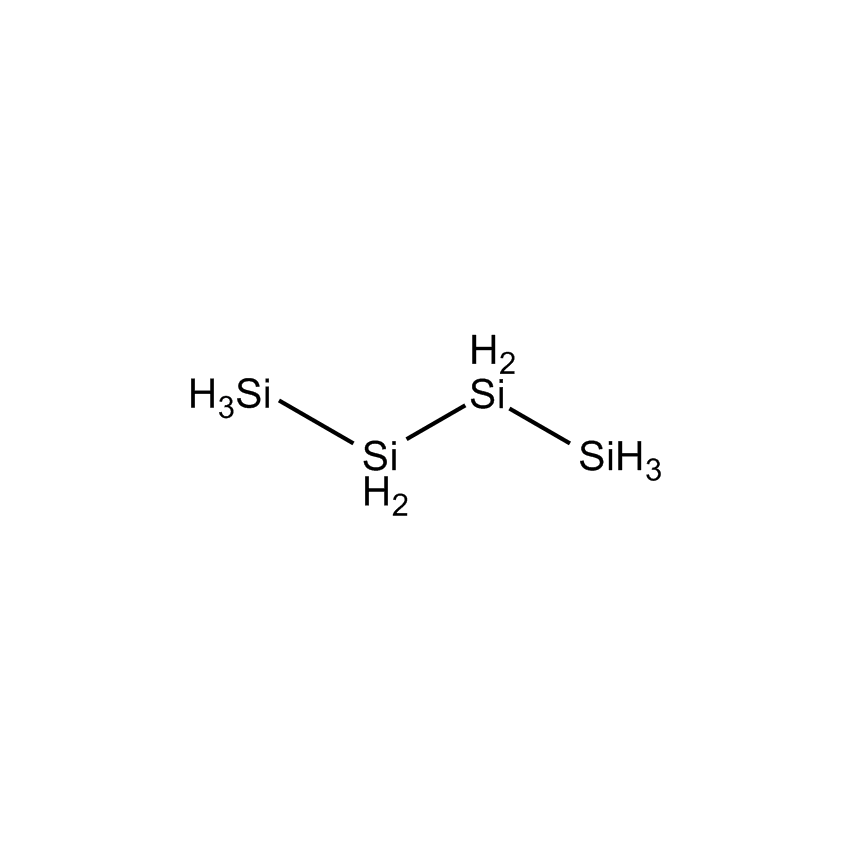
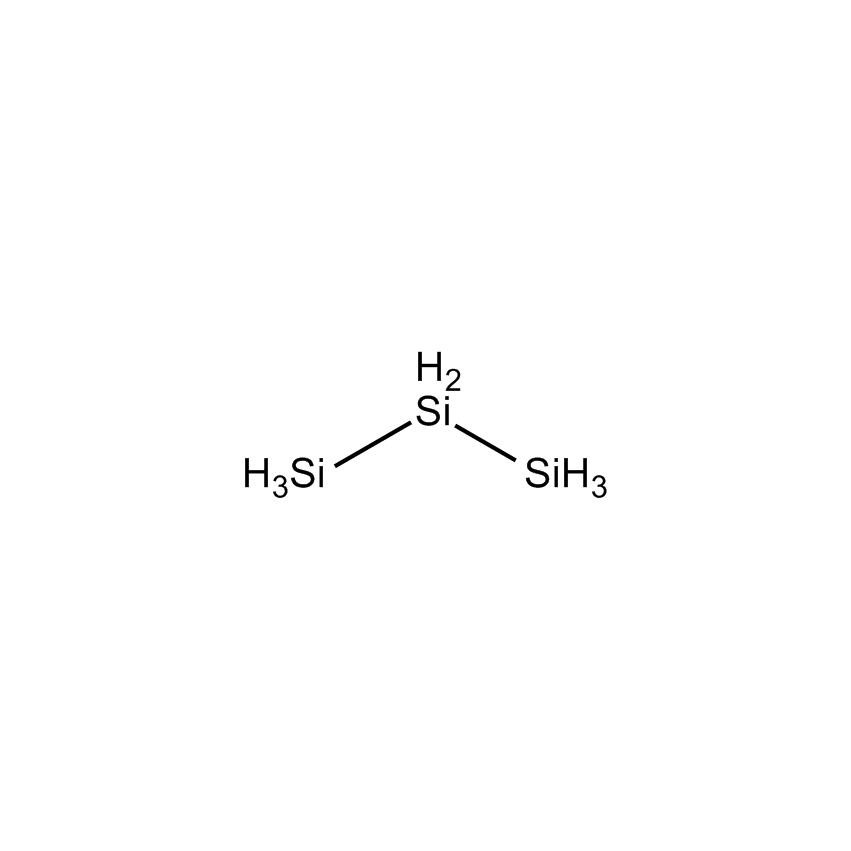
Perhydridooligosilanes differ from silane and disilane in a number of significant ways. As the number of silicon atoms increases beyond two, electrons are capable of sigma-sigma bond conjugation. The consequences are that the optical absorption of oligosilanes shifts to longer wavelengths with additional silicon atoms. Apart from the obvious advantage oligosilanes possess in CVD by having a greater number of silicon atoms than silane or disilane, the dissociative adsorption of two of the three hydrogen atoms on terminal silicon atoms has a lower energy barrier. This significantly reduces the thermal budget for deposition. While silane and disilane undergo relatively low rates of deposition below 850 °C, trisilane and higher linear oligosilanes have demonstrated practical deposition rates as low as 600 °C. Branched silanes, such as isotetrasilane and neopentasilane, undergo deposition at lower temperatures than their linear analogs, with practical deposition rates at temperatures as low as 450 °C. At atmospheric pressure, isotetrasilane and neopentasilane maintain liquid behavior as high as 100 °C, allowing on-substrate photolytic, electron-beam, or laser-induced conversion to silicon-rich films.
Volatile Higher Silanes
| SKU | Image | Name |
|---|---|---|
| SII6463.4 |
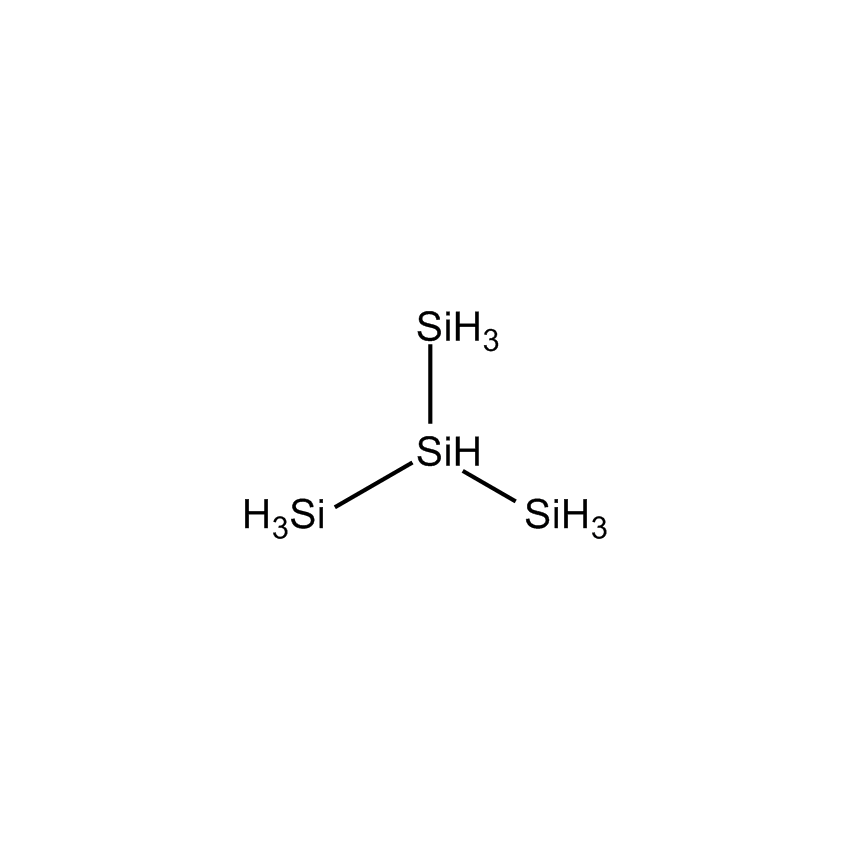
|
ISOTETRASILANE |
| SIN6597.07 |
|
NEOPENTASILANE |
| SIT7880.0 |
|
n-TETRASILANE |
| SIT8709.6 |
|
TRISILANE |
Oligomethylsilanes and polymethylsilanes undergo conversion to carbosilanes at temperatures above 650 °C.