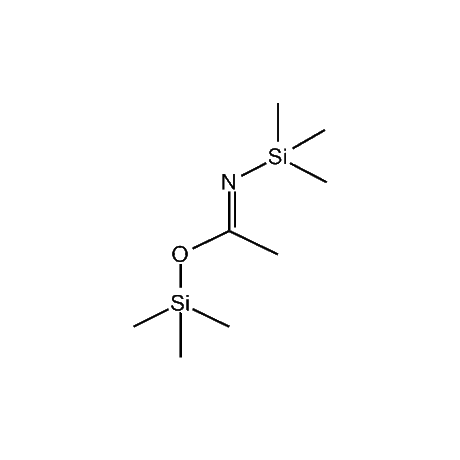
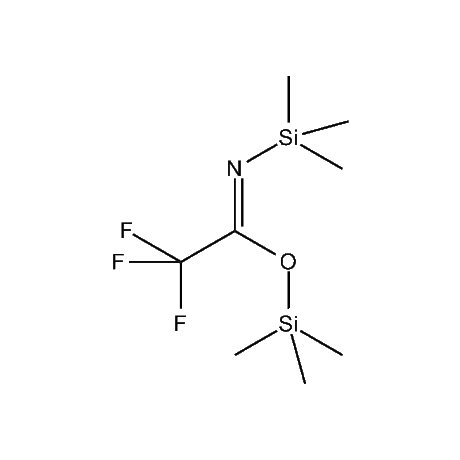
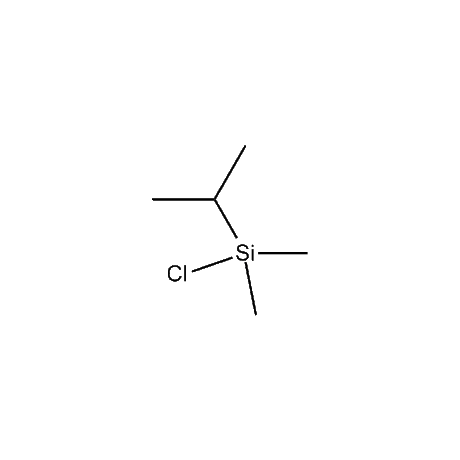
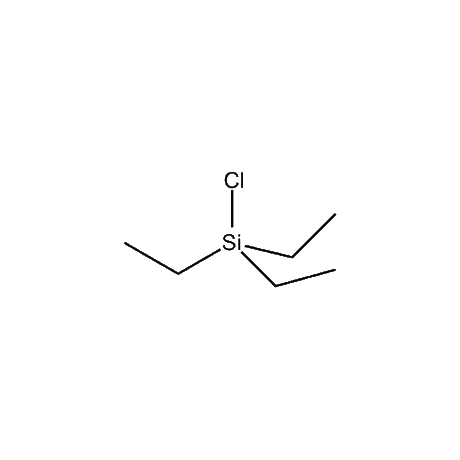
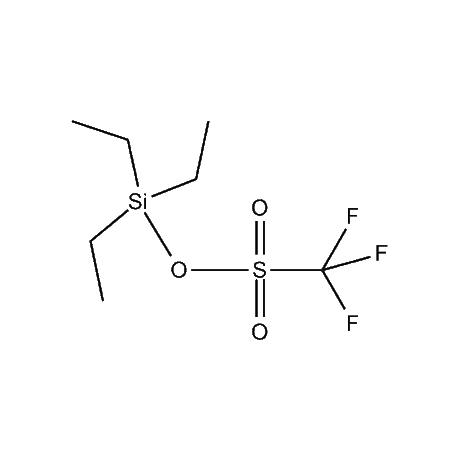
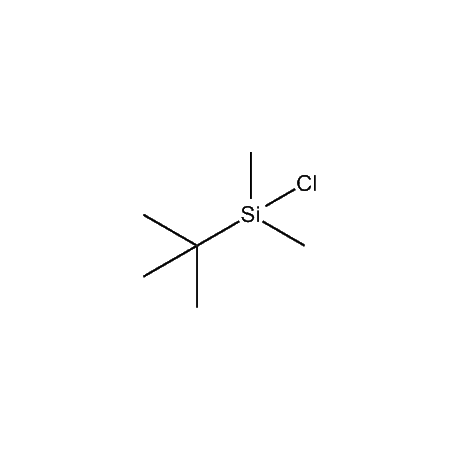
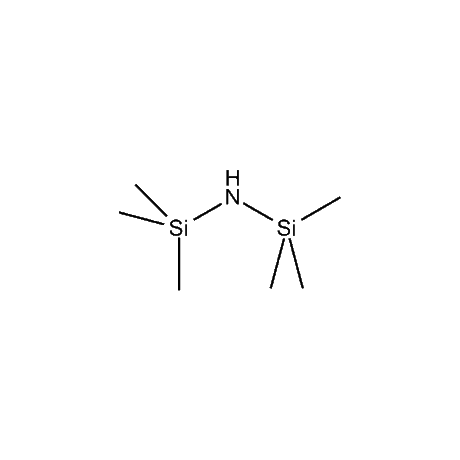
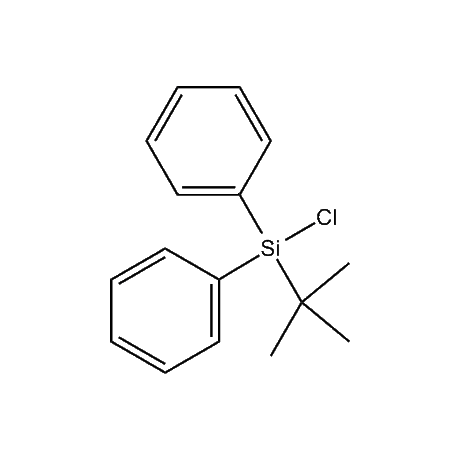
It is very common that in a multi-step synthetic sequence the protection of one or more functional groups will be required. The most common groups that fall into this class are alcohols, amines, thiols, acids, and carbonyls, in particular ketones and aldehydes. For the protection of functional groups with active hydrogens, such as alcohols and amines, organosilanes have proven to be among the best and most favored alternatives.1 The reasons for this are that the silyl groups can be both introduced and removed in high-yield, facile processes. In addition, the organosilanes have the ability to be modified both sterically and electronically, thus giving them excellent selectivity and versatility in their reactivity, stability, and ease of deprotection. Indeed, in many of the more complicated and lengthy synthetic sequences it is common to have a requirement to remove one organosilyl-protected group in the presence of other similar groups. A thorough presentation on the selective deprotection of one organosilyl-protected group in the present of another has been compiled.2 Gelest offers an extensive range of organosilanes designedfor the protection of various functional groups including alcohols, diols, amines, thiols, carboxylic acids and terminal alkynes. Of particular note is the use of the trimethylsilylenol ether of acetone, SII6264.0,for the protection of diols as their acetonides in a very fast and high-yield reaction.3 Gelest is proud to offer the E. J. Corey BIBS reagent for the protection of alcohols, amines, and carboxylic acids and the formation of highly stable enol silyl ethers.4
References:
1. Wuts, P. G. M.; Greene, T. W. “Greene’s Protective Groups in Organic Syntheses” Wiley and Sons, 2007.
2. Larson, G. L. “Silicon-Based Blocking Agents”Gelest, Inc. 2014.
3. Larson, G. L.; Hernández, A. J. Org. Chem. 1973, 38, 3935.4.Liang, H.; Hu, L.; Corey, E. J. Org. Lett. 2011, 13, 4120
4. Liang, H.; Hu, L.; Corey, E. J. Org. Lett. 2011, 13, 4120