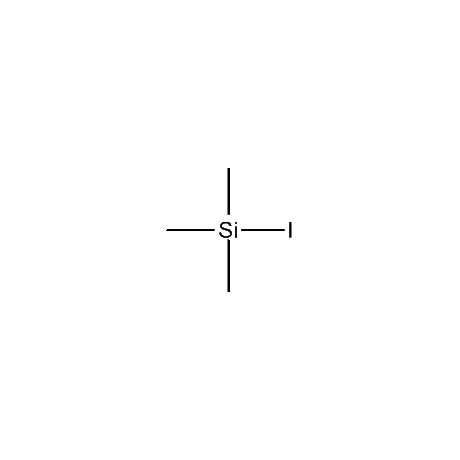Organosilane reductions are very commonplace in synthetic organic chemistry with the Si-H bond, being weakly polarized such that the hydrogen is hydridic in nature. Because the Si-H bond is only weakly hydridic, the silanes have the potential to be highly selective reducing agents, reducing a number of functionalities with excellent tolerance for other reducible functionalities. Organosilanes have further advantages in terms of low toxicity, ease of handling, and ease of final product purification. In addition, a number of enantioselective organosilane reductions have been reported. Ionic and organometalic-catalyzed organosilane reductions have been extensively reviewed.1,2
The extensive electronic and steric diversity of organosilanes offers a large range of selectivities in reactivity. For example, the homopolymeric methylhydrogensiloxane, HMS-992, and related homopolymers represent a high molecular weight silane reducing agent that can offer significant product isolation advantages.3 Diphenylsilane, SID4559.0, has been shown to selectively reduce amides to aldehydes.4 Triisopropylsilane, SIT8385.0, has been shown to offer a steric advantage leading to a higher degree of the β-aryl glucoside from the hemiketal precursor.5
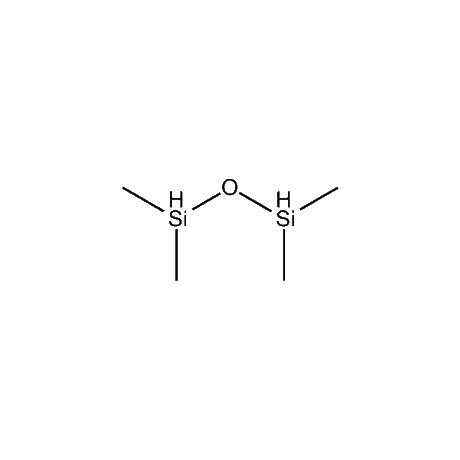
Tetramethyldisiloxane, TMDS, SIT7546.0, has recently been shown to be highly useful in the reduction of hemiketals to the corresponding ether. This has been applied to successful syntheses of the pharmaceutically important gliflozin family of diabetes 2 Specific Glucose Transport 2 (SGLT2) inhibitor drugs, such as canagliflozin, (Invokana) and dapagliflozin (Farxiga) among others.6 TMDS has also been shown to very effectively reduce nitroaryls to anilines7 and is an improvement over triethylsilane in the reduction of a ketone to a methylene in the production of a key intermediate in the preparation of ziprasidone.8
References:
1. Larson, G. L.; Fry, J. L. “Ionic and Organometallic-Catalyzed Organosilane Reductions”, Organic Reactions, Vol. 71, 2008, Denmark, S. E. Ed. Wiley and Sons, Hoboken, NJ.
2. Larson, G. L. “Silicon-Based Reducing Agents” Version 2.0, Gelest, Inc. 2008.
3. Chandrasekhar, S.; Reddy, Ch. R.; Ahmed, M. Synlett. 2000, 1655.
4. Bower, S.; Kreutzer, K. A.; Buchwald, S. L. Angew. Chem. Int. Ed. 1996, 35, 1515.
5. Elsworth, B. A. et al. Tetrahedron: Asymmetry 2003, 14, 3243.
6. a. Lemaire, S. et al. Org. Lett. 2102, 14, 1480. b. Nomura, S. et al. J. Med. Chem. 2010, 53, 6355. c. Lee, J. et al. Bioorg. Med. Chem. 2010, 18, 2178.
7. Nagashima, H. et al. J. Am. Chem. Soc. 2009, 131, 15032.
8. Nadkarni, D.; Hallissey, J. F. Org. Proc. Res. Dev. 2008, 12, 1142.
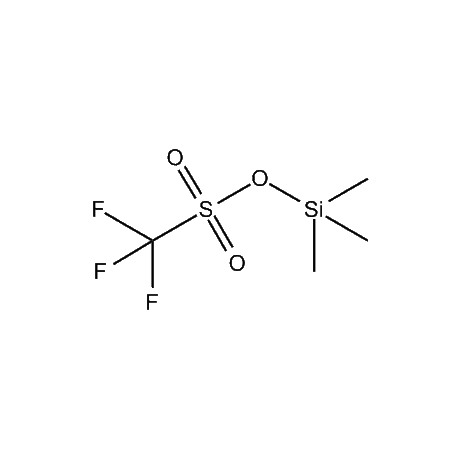
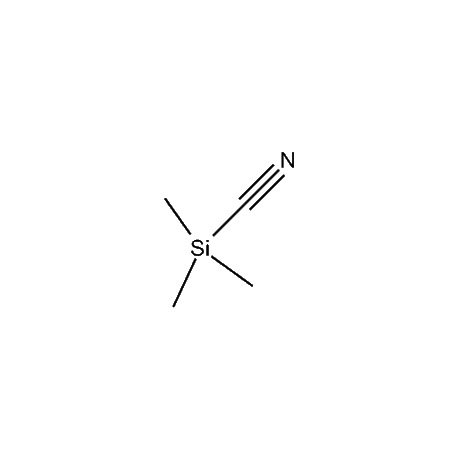
Gelest offers a number of other organosilane reagents that are of high use in the organic synthetic area. These include, among others, trimethyliodosilane, trimethylbromosilane, trimethylsilyl triflate, cyanotrimethylsilane, a number of silyl enol ethers and silyl ketene acetals, in addition to an extensive variety of organosilanes that are attached to inorganic surfaces for the purpose of final purification of desired products. Gelest also offers several non-silicon-based synthetic reagents that complement and, oftentimes, assist in the reactions of the silicon-based reagents. These include a number of Lewis acid catalysts such as tin tetrachloride, tetraisopropyltitanate, tris(pentafluorophenyl)boron, and a variety of metal diketonates. In addition Gelest offers several metal-organic reagents for various synthetic transformations.